 | |
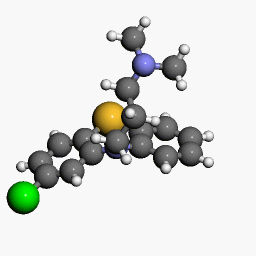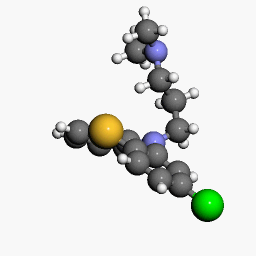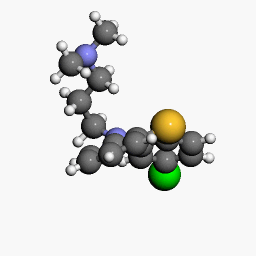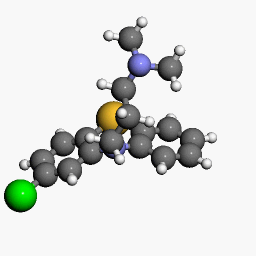
| Chlorpromazine |
There is a running debate over whether a drug needs to be clean or dirty in order to be effective. The debate has been brought into sharp focus by three decades of targeted drug discovery in which selective, (relatively) clean drugs hitting single proteins have led to billion dollar markets and relief for millions of patients. For instance consider captopril which blocks the action of angiotensin-converting-enzyme (ACE). For a long time this was the world's best-selling blood pressure-lowering drug. Similar single drug-single protein strategies have been effective for other major diseases like AIDS (HIV protease inhibitors) and heart disease (HMG-CoA inhibitors like Lipitor).
However recent thinking has veered in the direction of drugs that are "selectively non-selective". The logic is actually rather simple. Most biological disorders are modulated by networks of proteins spanning several physiological systems. While some of these are more important than others as drug targets, there are sound reasons to think that targeting a judiciously picked set of proteins rather than just a single one would be more effective in treating a disease. The challenge has been to purposefully engineer this hand-picked target promiscuity into drugs; mostly it is found accidentally and in retrospect, as in case of the anticancer drug Gleevec. Since we couldn't do this rationally (it's hard even to target a single protein rationally), the approach was simply to test different drugs without worrying about their mechanism and let biology decide which drugs work best. In fact, in the absence of detailed knowledge about the molecular targets of drugs this became a common approach in many disorders, and even today the FDA does not necessarily require proof of mechanism of action for a drug as long as it's shown to be effective and safe. In psychiatry this has been the status quo for a long time.
But now it looks like this approach has run into a wall. Lack of knowledge of the mode of action of psychiatric drugs may have led to accidental discovery, but the NIH thinks that it has also stalled the discovery of original drugs for decades. The agency has now taken note of this and as a recent editorial in Nature indicates, they are now going to require proof of mechanism of action for new psychiatric medicines. The new rules came from an appreciation of ignorance:
"Part of the problem is that, for many people, the available therapies simply do not work, and that situation is unlikely to improve any time soon. By the early 1990s, the pharmaceutical industry had discovered — mostly through luck — a handful of drug classes that today account for most mental-health prescriptions. Then the pipeline ran dry. On close inspection, it was far from clear how the available drugs worked. Our understanding of mental-health disorders, the firms realized, is insufficient to inform drug development."
For several years, the NIMH has been trying to forge a different approach, and late last month institute director Thomas Insel announced that the agency will no longer fund clinical trials that do not attempt to determine a drug or psychotherapy’s mechanism of action. Without understanding how the brain works, he has long maintained, we cannot hope to know how a therapy works."This is a pretty significant move on the part of the NIMH since as the article notes, it could mean a funding cut for about half of the clinical trials that the agency is currently supporting. The new rules would require researchers to have much better hypotheses regarding targets or pathways in the brain which they think their therapies are targeting, whether the therapies are aimed at depression or ADD. So basically now you cannot just take a small molecule that seems to make mice happier and pitch it in clinical trials for depression.
Personally I have mixed feelings about this development. It would indeed be quite enlightening to know the mechanism of action for neurological drugs, and I will be the first one to applaud if we could neatly direct therapies at specific patient populations based on known differences in signaling pathways for instance. But the fact remains that our knowledge of the brain is still too primitive and clunky for us to easily formulate target-based hypotheses for new psychiatric drugs. For complex, multifactorial diseases like schizophrenia there are still dozens of hypotheses for mechanisms of action. In addition there is a reasonable argument that it's precisely this obsession with targets and mechanisms of action that has slowed down pharmaceutical development; the thinking is that hitting well-defined targets has been too reductionist, and many times it doesn't work because it disregards the complexities of biology. If you really wanted to discover a new antidepressant, then it really might be better to look at what drug makes mice happier rather than try to design drugs to hit specific protein targets that may or may not be involved in depression.
So yes, I am skeptical about the NIMH's new proposal, not because an understanding of mechanism of action is futile - it's the opposite, in fact - but because our knowledge of drug discovery and design is still not advanced enough for us to formulate and successfully test target-based hypotheses for complex psychiatric disorders. The NIH thinks that its approach is necessary because we haven't found new psychiatric drugs for a while, but in the face of biological ignorance what it may do might be to make the situation worse. I worry that requiring this kind of proposal would simply slow down new psychiatric drug discovery for want of knowledge. Perhaps there is a middle ground in which you require a few trials to demonstrate mechanism of action while allowing the majority to proceed on their own merry way, simply banking on the messy world of the biology to give them the answer. Biology is too complex to be held hostage to rational thinking alone.
Good thing the FDA approves drugs and not the NIH. So some academic researches will lose funding for clinical trials, this does not change the big picture at all.
ReplyDeleteSurely the success rate for clinical trials funded by NIMH is (like elsewhere) pretty low? Biomarker validation seems to be in general a wide valley of death, so I would have to assume that some good will come out of requiring people to yield scientific knowledge in the likely event that the proposed therapy doesn't work. Plus it would seem to go towards what academic researchers should be better at, which is generating knowledge rather than therapies.
ReplyDeleteGood point.
Delete